Symbol Glossary
# | Symbol | Symbol Title | Symbol Description | Standard Reference | Standard |
|---|---|---|---|---|---|
1.0 |  | Manufacturer | Indicates the medical device manufacturer | 5.1.1; Annex A @ A.2 | ISO 15223-1:2021 |
2.0 |  | Authorised representative in the European Community /
European Union
|
Indicates the authorized representative in the European Community / European Union | 5.1.2; Annex A @ A.4 | ISO 15223-1:2021 |
3.0 |  | Date of Manufacture | Indicates the manufacturer’s catalogue number so that the medical device can be identified | 5.1.3; Annex A @ A.5 | ISO 15223-1:2021 |
4.0 |  | Use-by Date | Indicates the date after which the medical device is not to be used | 5.1.4 | ISO 15223-1:2021 |
5.0 |  | Batch Code
(Lot Number)
| Indicates the manufacturer’s batch code so that the batch or lot can be identified | 5.1.5; Annex A @ A.7 | ISO 15223-1:2021 |
6.0 |  | Catalogue Number | Indicates the manufacturer’s catalogue number so that the medical device can be identified | 5.1.6; Annex A @ A.8 | ISO 15223-1:2021 |
7.0 | 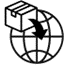 | Importer | Indicates the entity importing the medical device into the locale | 5.1.8; Annex A @ A.10 | ISO 15223-1:2021 |
8.0 |  | Distributor | Indicates the entity
distributing the medical device into the locale
| 5.1.8; Annex A @ A.11 | ISO 15223-1:2021 |
9.0 |  | Sterilized using Ethylene Oxide | Indicates a medical device that has been sterilized using Ethylene Oxide (EO) | 5.2.3 | ISO 15223-1:2021 |
10.0 |  | Sterilized using irradiation | Indicates a medical device that has been sterilized using irradiation | 5.2.4 | ISO 15223-1:2021 |
11.0 |  | Do not Resterilize | Indicates that the medical device is not to be resterilized
Note: This symbol is only to be used when there is an accompanying sterile symbol
(#9, #10)
| 5.2.6 | ISO 15223-1:2021 |
12.0 |  | Non-Sterile | Indicates that the medical device has not been subjected to a sterilization process
Note: This symbol should only be used to distinguish between identical or similar medical devices sold in both sterile and non-sterile conditions. | 5.2.7 | ISO 15223-1:2021 |
13.0 |  | Do Not Use If Package is Damaged and consult IFU | Indicates that a medical device that should not be used if the package has been damaged or opened and that the user should consult the IFU for additional information
Note1: This symbol can also mean “Do not use if the product sterile barrier system or its packaging is compromised”.
Note2: For products that do not have IFU, the recommendation to consult them does not apply. | 5.2.8 | ISO 15223-1:2021 |
14.0 |  | Single Sterile Barrier System | Indicates a single sterile barrier system
Note: This symbol shall be placed adjacent to or in combination with symbol #9 or #10.
| 5.2.11; Annex A @ A.22 | ISO 15223-1:2021 |
15.0 |  | Double Sterile Barrier System | Indicates two sterile barrier systems.
Note: This symbol shall be placed adjacent to or in combination with symbol #9 or #10.
| 5.2.12; Annex A @ A.22 | ISO 15223-1:2021 |
16.0 |  | Single Sterile Barrier System with Protective Packaging Inside | Indicates a single sterile barrier system with protective packaging inside
Note: This symbol shall be placed adjacent to or in combination with symbol #9 or #10.
| 5.2.13; Annex A @ A.22 | ISO 15223-1:2021 |
17.0 |  | Single Sterile Barrier System with Protective Packaging Outside | Indicates a single sterile barrier system with protective packaging outside
Note: This symbol shall be placed adjacent to or in combination with symbol #9 or #10.
| 5.2.14; Annex A @ A.22 | ISO 15223-1:2021 |
18.0 |  | Fragile, Handle with Care | Indicates a medical device that can be broken or damaged if not handled carefully | 5.3.1 | ISO 15223-1:2021 |
19.0 | 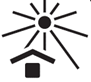 | Keep Away from Sunlight | Indicates the medical device that needs protection from light sources
Note: This symbol can also mean “Keep away from heat”.
| 5.3.2 | ISO 15223-1:2021 |
20.0 |  | Keep Dry | Indicates a medical device that needs to be protected from moisture | 5.3.4 | ISO 15223-1:2021 |
21.0 |  | Temperature Limit | Indicates the temperature limits to which the medical device can be safely exposed | 5.3.7 | ISO 15223-1:2021 |
22.0 |  | Do not re-use | Indicates that the medical device is intended for one single use only | 5.4.2 | ISO 15223-1:2021 |
23.0 |  | Consult IFU | Indicates the need for the user to consult the instructions for use | 5.4.3 | ISO 15223-1:2021 |
24.0 |  | Consult eIFU | Indicates the need for the user to consult the instructions for use in electronic format
Note: The eIFU indicator can be a manufacturer’s website URL or some other appropriate indication that the instructions for use are available in an electronic format.
| Annex A @ A.16 | ISO 15223-1:2021 |
25.0 |  | Caution | Indicates that caution is necessary when operating the device or control close to where the symbol is placed, or that the current situation needs operator awareness or operator action in order to avoid undesirable consequences | 5.4.4 | ISO 15223-1:2021 |
26.0 |  | Contains or Presence of Natural Rubber Latex | Indicates the presence of dry natural rubber or natural rubber latex as a material of construction within the medical device or the packaging of the medical device
Note: This symbol is intended to warn those people who can have allergic reactions to certain latex proteins.
| 5.4.5 | ISO 15223-1:2021 |
27.0 |  | Does Not Contain or No Presence of Natural Rubber Latex | Indicates that the medical device does not contain dry natural rubber or natural rubber latex as a material of construction within the medical device or the packaging of the medical device | N/A | N/A |
28.0 | 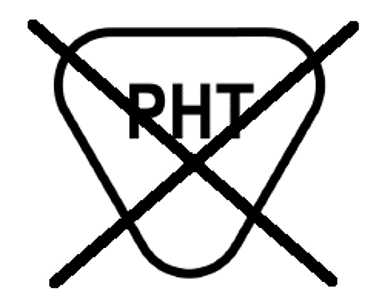 | Does Not Contain or No Presence of Phthalate | Indicates that the medical device does not contain or has no presence of phthalate as a material of construction within the medical device or the packaging of the medical device | N/A | N/A |
29.0 |  | Single Patient Multiple Use | Indicates a medical device that may be used multiple times (multiple procedures) on a single patient | 5.4.12 | ISO 15223-1:2021 |
30.0 |  | In Vitro Diagnostic Medical Device | Indicates a medical device that is intended to be used as an in vitro diagnostic medical device | 5.5.1 | ISO 15223-1:2021 |
31.0 |  | Non-Pyrogenic | Indicates a medical device that is non-pyrogenic | 5.6.3 | ISO 15223-1:2021 |
32.0 |  | Medical Device | Indicates the item is a medical device | 5.7.7 | ISO 15223-1:2021 |
33.0 | CE Registration Mark | The CE marking:
shows that ITLAP has checked that its products meet EU safety, health or environmental requirements, is an indicator of a product’s compliance with EU legislation and allows the free movement of products within the European market | N/A | N/A | |
34.0 |  | CE0123 Registration Mark | Indicates that TÜV SÜD was the Notified Body involved in conformity assessment | N/A | N/A |
35.0 |  | CE0197 Registration Mark | Indicates that TUV Rheinland was the Notified Body involved in conformity assessment | N/A | N/A |
36.0 |  | Prescription Only | Caution: U.S. Federal Law restricts this device to sale by or on the order of a physician | N/A | N/A |
37.0 | Malaysia Medical Device Registration Number | A registered medical device shall be labelled with Malaysian Medical Device Registration Number | N/A | N/A | |
38.0 |  | Package Quantity | Indicates the number of units contained in the package | N/A | N/A |
39.0 |  | Carton Weight | Indicates the weight of the carton | N/A | N/A |
40.0 |  | Contains or Presence of Phthalate | This symbol shall be accompanied by the abbreviated designation of the particular phthalate(s) used. The particular phthalate(s) designation shall be located adjacent to the symbol. | 4.2 | EN 15986:2011 |